Plant thinking in medicine - renal immunology in native and grafted kidney. Immune therapy for Glomerulonephritis
____________________________________
Contents
(i) Lymphocyte, origin, migration & distribution
(ii) Activities related to immunology
Section A: Renal immunology
- Immunology of native and transplant kidney,
- Types of immuno-therapy.
- Immunoglobulin,
- Polyclonal,
- Orthoclonal,
- Monoclonal **.
- Small molecules
- CAR-T Immuno-therapy
- Puzzle game in the cartoon
** (Immune check point inhibitors under Monoclonal section)
Section B: Immunological management of Glomerulonephritis
- Induction therapy for remission
- Maintenance therapy for GN in remission
- Adjuvant therapy
- Prophylaxis
- Treatment of Relapse cases
_____________________________________
(i) Lymphocytes origin, migration and distribution
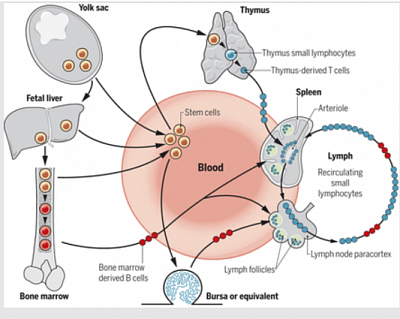
Origin and migration of T and B cells:
(Reprinted with permission from the Royal Society, London)
Hemopoietic stem cells originating in the yolk sac, fetal liver, or bone marrow migrate through the blood to the thymus, where they differentiate into T lymphocytes. Others differentiate within the bone marrow in mammals, or the bursa of Fabricius in birds, to produce B cells. Mature T and B cells circulate in the blood and lymph and colonize the T and B cell compartments of the lymphoid tissues.
____________________________________
(ii) Activities in relation to immunology
Section A: Renal immunology
Contents
- Immunology of native and transplant kidney,
- Types of immuno-therapy
- Immunoglobulin,
- Polyclonal,
- Orthoclonal,
- Monoclonal.
- Small molecules
- CAR-T Immuno-therapy
- Puzzle game in the cartoon
____________________________________
(ii) Activities in relation to immunology:
Section A
A1. Immunology, native & graft
🌿 Plants do not have central nervous system, but they react to pain, irritation, heat, touch, light and some times sound. They do it by chemical substances. In human, immune system is a response system without being a neuronal system. In response to auto or hetero antigenic stimuli, activated T-lymphocytes react with B-lymphocytes and an array of antigenic upregulation, monoclonal antibody production, complement activation and multitudes of inflammatory responses are formulated culminating in either diseases or tolerance.
Translational medicine manipulates this non-neuronal system in producing monoclonal antibodies against key molecules, cell membrane receptors and antigens, complement products, inflammatory products, activating factors, idiotypic antibodies etc, targeting key molecules of the disease process.
Therapeutic Mabs are produced in vertebrates and humanized to avoid activation of ideotypic/antiideotypic responses.
____________________________________
A2. Types of immunotherapy:
- Immunoglobulin
- Small molecules
- Cellular: CAR-T
____________________________________
A3. Immonoglobulins
In general three kinds of immunoglobulins:
- Polyclonal
- Orthoclonal
- Monoclonal (-mab)
____________________________________
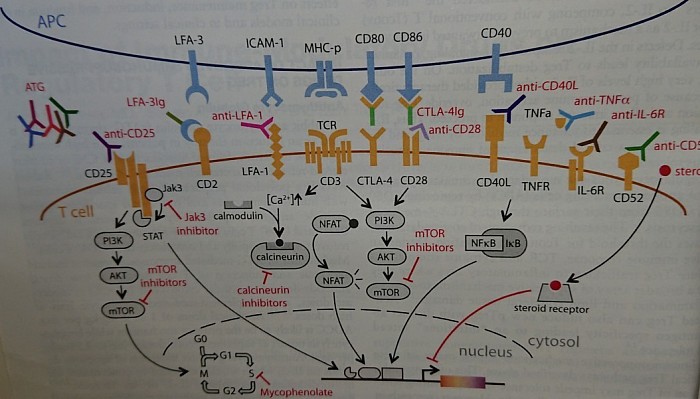
A4. Polyclonal immunoglobulin
- Thymoglobulin/ Antithymocyte globulin (Atgam) from rabbit/ horse serum after inoculation with thymocytes (72% T-lymphocytes, 8% B-lymphocytes). T-lymphocyte depleting.
A polyclonal immunoglobulin against mostly T-lymphocytes used in transplant induction (ABO-c with high PRA/Crossmatch+ cases, ABO-i) and treatment of Type 2a onward acute T-cell mediated rejection. Follow local practice. You need to use CMV and PCP prophylaxis and monitor for CMV & BKV serology every 1-3 months. Shortterm adverse effects are sepsis and cytokine release syndrome while longterm are CMV, BKV, EBV, PVB19 related disease and hematological and other malignancies.
- IV Immunoglobulin-G.
This human IgG is derived from pooled plasma. It contains multitude of antibodies, ideotypic antibodies, antiideotypic antibodies and antiantiideotypic antibodies against various antigens, virus, antibodies.
Thats why it's used in immunodeficiency diseases, treatment of viral disease (BKV, pvb19, CMV), audio immune disease (ITP, DAT +ve haemolytic anaemia, Lupus etc), Crossmatch positive renal transplant, high PRA transplant, ABO-i transplant with cascade plasmapharesis, AR with donor specific antibody (DSA) positive cases.
In borrowed kidney, some time we use at least 12 hours preceding Plasmapheresis in case of DSA positive cases for B cell mediated AR, specially in CMV positive recipients.
Other indications is CMV disease.
____________________________________
A5. Orthoclonal
Orthoclonal Antibody against CD3 membrane receptor of T3 lymphocytes (OKT3) that differentiate into T4 & T8 lymphocytes. It depletes T3 lymphocytes thereby depletes both T4 and T8 lymphocytes. Derived from mouse serum after inoculation with CD3 positive T lymphocytes.
Used as induction therapy in highly sensitized transplant and type 2a onward AR. We used this 20 years ago but ceased to use because of severe and some time fatal cytokine release syndrome aw pulmonary oedema.
Good to know that it is the first monoclonal antibody used in transplant.
____________________________________
A6. Monoclonal immunoglobulins ** (Immune check point inhibitors under this section, mentioned in Anti Tumor mab )
Approved (link for a summary table)
- a. Campath (humanized 1H).
Roy Calne, FRS, introduced liver transplant in UK after US. As transplant surgeon he produced Anti CD52 (the common marker of both B and T lymphocytes) monoclonal antibody in his Cambridge University pathology lab that has been being used in renal, liver and pancreatic transplant since then. Both B & T lymphocytes are depleted for at least 3-6 months after 2X20mg dose. Later new naive lymphocyte cell line develops that are tolerant to the graft.
Recipients are kept on low dose CNI with steroid free or low dose prednisolone. RCTs showed lesser AR and better longterm renal survival. Currently a few RCTs are ongoing in both renal and simultaneous pancreas and kidney transplantation.
Also used in CLL & some auto immune diseases to cause B & T lymphocytes depletion by cytotoxic t lymphocytes emeliorating CLL & attenuating immune disease. This is called Targetted cell destruction by t lymphocytes.
Dacluzimab and Basiliximab (trade name Simulect) are chimeric mouse-human monoclonal antibody to the α chain of the IL-2 receptor (CD25) of T cells. With 2X20 mg of Basiliximab at 4 weeks interval, it binds IL2 receptors (CD25) on T lymphocytes to block IL2 binding to it's receptors. Thus prevents activation and proliferation of t-cell and consequently down regulation of b-cell activation for antibody production. Thus prevent rejection.
Biological half-life: 7.2 days
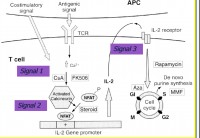
- c. Belatacept. T lymphocytes co-stimulatry signal inhibitor.
[ref: Mechanism of graft rejection & tolerance -signal 1, signal 2, signal 3]
This is a monoclonal antibody against
cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4), selectively inhibits T-cell activation through co-stimulation blockade. ((In immune response after graft surgery, antigenic stimulation of CD3 on t- lymphocytes leads to upregulation of T4 (CD4+) and T8 (CD8+) lymphocytes (Signal 1). This activation of t4 cells also needs simultaneously activated co-stimulatory receptor CTLA-4 of CD3 T lymphocytes by the corresponding CTLA-4 Ligand on APC (antigen presenting cells). This is known as signal-2. This leads upregulation of CD4 T lymphocytes resulting in IL2 production through calcinurin-calmodulin pathway. IL2 further stimulate proliferation of t4 lymphocytes through paracrine and autocrine pathway to release more IL2 and subsequently unregulate B- lymphocytes for antigraft antibodies. As such T cell activation is prevented by co-stimulation (signal 2) blockade by Belatacept. This is akin to graft tolerance. IL2 induces cell proliferation by promoting cell cycle at S phase (signal 3), that is blocked by mTOR inhibitor.
BENEFIT trial is on Belatacept.
- d. Rituximab.
Anti CD20 monoclonal anybody. It causes B lymphocytes depletion.
Used in resistant GN, LN and B lymphocyte mediated Antibody mediated transplant Rejection.
- e. Eculizumab
Now considered a panacea in immune therapy. A UK product, against activated complement 5 (C5a), component of lyric terminal complement complex in complement mediated cell lysis that happens in haemolytic Uremic syndrome, PAH, PNH, B cell mediated rejection of borrowed kidney and many other immune mediated inflammatory diseases. In CNI INDUCED HUS it is used with a possible cost of a million dollar annually.
You should know the standard treatment is Plasmapheresis and change to mTOR inhibitors.
You need to Vaccinate against N Meningococcus with 2 weeks of administration, because of propensity of fatal meningitis due to nature of mechanism of action.
- f. Belimumab
Belimumab (Benlysta) is an anti B-lymphocyte stimulator [BLyS] monoclonal antibody). It has been found to have beneficial effects on clinical and laboratory parameters in patients with active SLE. In addition, the number of B cells and serum IgM were reduced over time.
Belimumab was approved by the US Food and Drug Administration (FDA) for use in patients with active SLE who are autoantibody-positive and are receiving standard therapy, including corticosteroids, antimalarials, immunosuppressives, and NSAIDs.
- g. Atacicept
Atacicept is a TACI-Ig fusion protein that inhibits BLyS and a proliferation-inducing ligand. In early phase studies, atacicept was demonstrated to have biologic effects in patients with SLE, resulting in a dose-dependent reduction in B cells and immunoglobulin levels.[60]
- h. Anticytokine monoclonal antibody
Various anticytokine therapies have been proposed, including monoclonal antibodies directed against interferon-α, interleukin (IL)-1, IL-6, IL-10, tumor necrosis factor alpha (TNF-α, known as Infliximab) and Vascular endothelial growth factor mab, etc.
a. Infliximab is considered as a panacea for psoriasis. A guy moved with this along the eastern shore of USA erasing psoriasis on his journey through. We heard it from him here during his sponsored visit. It's been used in rheumatoid arthritis and similar condition.
(vascular endothelial grow factor monoclonal antibody, Bevacizumab) is mainly used in carcinomas for regression of tumor by inhibiting angiogenesis.
c. Denosumab works by targeting protein called RANKL which controls the activity of osteoclasts. This stops bone cells being broken down and strengthens the bone.
- i. Anti Tumor monoclonal antibody
i1. Target antigen targeting mab
Campath, Rituximab, are examples for treTing CLL and Lymphoma.
Cancer cell have peoteins on their surface. Monoclonal antibodies specific to this protein, when redbinds to this cancer antigenic protein, make the cancer cell susceptible to destruction by T lymphocytes.
i2. Immune checkpoint inhibitor
T cells contain proteins on the surface, which render them inactive in destroying other cells, when the other cells bind to this protein with the help of ligands, the t-cells then goes into anergy, or simply become inactive in destroying the cancer cells. This t-cell surface antigens are known as immmuecheckpoint signal. Mab against this antigens renders them active and they begin to destroy tumor cells.
- Belatacept mentioned above is anti CTLA-4 mab, when binds to it, makes it unavailable to bind to its ligand on the graft cells or APC's, thus makes the t-cell unable to destroy graft cells.
- Immunecheckpoint PD-1 on surfsce of t cell, when binds to PD-L1 on other cells or tumor cells, they become inactive and tumor cells grow unabated. Cancer cells have a lot of PD.
MAB against PD-1 or PD-L1 rendered them inactive, and make the t cells destroy the tumor cells.
_____________________________________
A7. Small molecules
Two types:
Tyrosine kinase inhibitors (-nib) and proteosome inhibitors (-mib).
These are small molecules that inhibit intracellular Proteosome enzymes (-mib) inhibiting break down of intracellular protein, and Tyrosine kinase enzyme (TKI) inhibitors (-nib) that inhibits inhibitors of nuclear factor kappa B (NFKB) lytic enzymes, thus promoting break down of NFKB, downregulating nuclear transcription of cell proliferation, thus enhancing cell death by Apoptosis.
- a1. Proteosome inhibitors (-mib)
Famous is Bortezumib used in multiple myeloma as Velcaid either in combination as VCD (Velcaid, cyclophosphamide & dexamthasone) or as a single agent.
- b2. Tyrosine kinase inhibitors (-nib):
Less famous are TKI Imatinib, Sunitinib (sutent) used in renal cell carcinoma, and other nibs used in animal carcinomas.
____________________________________
A8. Cellular CAR-T therapy
CAR T-cell therapy: CAR-T (chimeric antigen receptor T-cell). Patient’s T-lymphocytes are genetically engineered in the laboratory (transfacted by vector-virus containing gene for t-cell receptor, monoclonal against specific target cancer cell antigens, i.e. CD19 present only in B lymphocytes in case of treating B leukemia and lymphoma). These CAR-T cells will bind to cancer cells and kill them when reinfused to the patients blood stream.
How prepared:
Blood from a vein in the patient’s arm flows through a tube to an apheresis machine, which removes the white blood cells, including the T cells, and sends the rest of the blood back to the patient. Then, the gene for a special receptor called a chimeric antigen receptor (CAR) is inserted into the T cells in the laboratory. Millions of the CAR T cells are grown in the laboratory and then given to the patient by infusion. The CAR T cells are able to bind to an antigen on the cancer cells and kill them.
____________________________________
A9. Puzzle game in the cartoon.
Having known so much about the plant, help yourself to match the antibodies against their targeted antigens. This is an exercise and a mind game for your diligence.
Section B: Immunological management of Glomerulonephritis
Contents
- Induction therapy for remission
- Maintenance therapy for GN in remission
- Adjuvant therapy
- Prophylaxis
- Treatment of Relapse cases
_____________________________________
B1. Induction therapy
Exclude secondary GN first:
- DN- do renal biopsy if UPCR>1, if GN treat like GN & refer endocrinologist for glucose control.
- Do ANA, Anti DS-DNA, ANCA and other serology as indicated.
- Virus: do HBV, HCV, HIV and other serology.
- Malignancy: age>55 years, do Myeloma panel, tumor markers (CA125 & 19.9, CEA, Alphafetoprotein, PSA in male).
- If negative, treat as primary GN.
- Exclude sepsis by CXR, if indicated urine C/S, procalcitonin, CRP.
- Don't ignore occult bleeding, i. e. Low TS, HB etc.
____________________________________
Induction of remission:
Use of Remission inducing medicines depends on the clinical and histological presentations as follows:
1. Crescentic GN, and LN IV:
a) IV Methylprednisolone (MP): 0.5gm daily infusion for 3 days followed by oral prednisolone daily 60 mg for 4-6 weeks. Subsequent course depends on the response.
Opinion: if response - tapper and continue at alternate day by 10 mg each month until 10 mg, then (reduce further) continue for long time.
b) IV Cyclophosphamide (CyP): 500 mg/sqm monthly cycles following IV MP, depending on the histopathology and immunology.
Duration, 6 cycles followed by 3 additional cycles at 3 months intervals. Or Oral CyP 1.5-2 mg/kg for 9 - 12 months.
2. Membranous Nephroparhy, and LN-V
a) IV Rituximab is used when remission is not achieved by prednisolone alone or with a second agent like CsA, CyP or MMF.
Dosage is 375 mg/sqm every 4 weeks for 4 cycles along with MMF & Prednisolone.
3. Primary nephrotic syndrome (NS) or nephrotic range proteinuria (includes MCD)
a) Prednisolone:
Oral 60 mg in adults (1 mg/Kg in padiatric cases) for 4-6 weeks in nephrotic syndrome due to primary GN, followed by slow tapering to maintenance dose.
You can use daily or alternate day regime during Tapering until 10 mg EOD is reached. Then continue at your judgement. (mechanism of action)
b) Cyclosporin-A, Cyclophosphamide or Mycophenolate mofetil (CsA, CyP or MMF) are used in non-response.
- Cyclosporin-A (CsA) is avoided in renal impairment. Desired trough level of CsA is 75-100 nmol/l. NS relapses commonly after withdrawal.
- Mycophenolate mofetil (MMF) is a safe and effective option works in most types of primary GN.
c) Budenoside (BUD)
Peyer’s patches located within the gut-associated lymphoid tissue have been identified as a major source of poorly O-galactosylated immunoglobulin A (IgA)1, which triggers the formation of nephritogenic immune complexes in IgA nephropathy (IgAN).
targeted-release formulation of budesonide (NEFECON), designed to deliver budesonide (BUD) to the Peyer’s patches in the ileum.
BUD rapidly metabolized by liver, hence is locally active, not systemically available.
Hence, active in chrohns disease and IGAN.
4. Acute nephritis - non nephrotic type
Most of the time you don't have to use immune therapy. It depends on degree of Proteinuria, serum creatinine and biopsy report.
In diffuse proliferative GN, FSGS and MGN with aggressive proteinuria or elevated creatinine, you need to start 4 to 6 weeks course of prednisolone 60 mg daily.
In case of response, continue at a slow tapering dose as above.
Otherwise, you can add a second agent for another 4 weeks to see response.
If there is response, treat as such. Discontinue otherwise.
_____________________________________
B2. Maintenance of remission
Prednisolone: low daily or EOD dose for long time (many nephrologists think that there is a low grade hypoadrenal state in GN cases).
CsA/FK, CyP, MM: either one may continue for 1 - 2 years,
Then changed to Azathioprine.
_____________________________________
B3. Adjuvant therapy
It is used in ANCA associated Crescentic GN (Clinical RPGN), Crescentic GN, non-ANCA with clinical RPGN and in some cases of FSGS cellular varient with RPGN presentation.
- IV IgG is used in certain situations of non-response like RPGN, class IV LN and collapsing FSGS. This is used with or without plasmapharesis.
- Hydroxichloroquin in SLE: This reduces need for higher prednisolone dosage during maintenance phase. It also reduces rates of relapse.
- Bactrim is used in Wageners Granulomatosis for a year. Some nephrologists think that low grade infection may be responsible for relapse.
_____________________________________
B4. Prophylaxis (Ref- CYCLOPS study design)
- Gastric protection: ranitidine or omeprazole, not cimetidine or misoprostol, for 6 months.
- Pneumocystis carinii pneumonia: avoid unless strong immunocompromized state, sulfamethoxazole/ trimethoprim 240 mg three times a week, or monthly aerosolised pentamidine may be used in Bactrim allergy, Stop at 3 months.
- Fungal infection: oral fluconazole, nystatin or amphotericin, for 12 weeks.
- Valgancyclovir or Acyclovir, when indicated.
- Bone protection: calcium and vitamin D tablets to those above 50 years age.
_____________________________________
B5. Relapse after remission
Treatment would be mostly same as for initial remission.
_____________________________________