ANCA associated Vasculitis (AAV)
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitides (AAV)
Contents
- Classification of vasculitis
- Nutshell
- Atlas of vasculitis
- Other vasculitis
- Pathophysiology
- Aetiologic factors
- Management
- Investigations
- Disease specific management
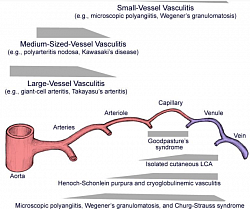
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitides (AAV) include Wegener Granulomatosis, Microscopic Polyangiitis, Churg-Strauss Syndrome and Renal-limited Vasculitis (pauci-immune).
ANCA-negative primary small-vessel vasculitides include Henoch-Schonlein Purpura, Essential Cryoglobulinemic Vasculitis, Kawasaki Syndrome and Cutaneous Leucocytoclastic Angiitis.
___________________________________
1. The EULAR and PRES Classification of Vasculitis
Consensus conference in 2005, endorsed by Paediatric Rheumatology European Society (PRES) and the European League against Rheumatism (EULAR).
1. Large vessel vasculitis:
- Takayasu arteritis.
2. Medium-sized vessel vasculitis:
- Childhood polyarteritis nodosa
- Cutaneous polyarteritis
- Kawasaki disease
- Primary vasculitis of the CNS, angiography-positive primary angiitis of the CNS (PACNS)
3. Small vessel vasculitis two types:
- Granulomatous and
- Nongranulomatous.
a) Granulomatous includes the following:
- Granulomatosis with polyangiitis GPA (formerly known as Wegener granulomatosis)
- Churg-Strauss syndrome
b) Nongranulomatous includes the following:
- Microscopic polyangiitis
- Henoch-Schönlein purpura
- Isolated cutaneous leukocytoclastic vasculitis
- Hypocomplementemic urticarial vasculitis
- Primary vasculitis of the CNS, angiography-negative, small vessel PACNS
Other vasculitides includes the following:
- Behçet disease
- Antiglomerular basement membrane (GBM) antibody disease
- Vasculitis secondary to infection (including hepatitis B associated polyarteritis nodosa), malignancies, and drugs, including hypersensitivity vasculitis
- Vasculitis associated with connective tissue diseases
- Cogan syndrome
- Unclassified
_____________________________
2. Nutshell
- ANCA is strongly associated for myeloperoxidase-ANCAs than for proteinase-3-ANCAs.
- Neutrophils, complement and effector T cells are also involved in AAV pathogenesis.
- With respect to treatment of AAV, glucocorticoids, cyclophosphamide and other conventional therapies are commonly used to induce remission in generalized disease.
- Pulse intravenous cyclophosphamide is equivalent in efficacy to oral cyclophosphamide but seems to be associated with less adverse effects.
- Methotrexate is used as a less-toxic alternative to cyclophosphamide to induce remission in non-organ-threatening or non-life-threatening AAV.
- Rtuximab is equally as effective as cyclophosphamide for induction of remission in AAV .
- Avacopan - specific orally-administered, selective complement C5a receptor inhibitor working on immune effector cells and molecules are being therapeutically targeted have been found promising.
______________________________
Classification, aetiology, pathogenesis and classic clinical features of AAV
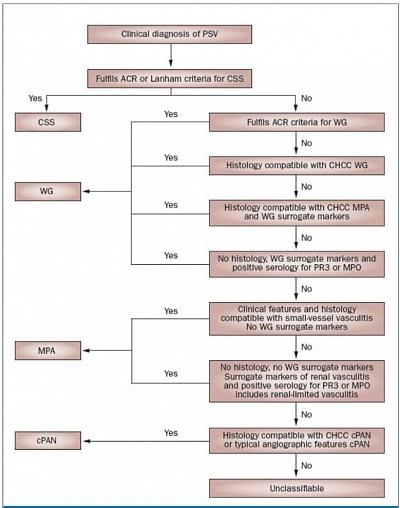
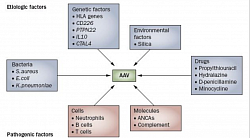
Primary Vasculitis classification (Picture de-classico): inflammation of blood vessels, causing wall thickening, stenosis, and occlusion with ischemia. Necrotizing inflammation that destroy segments of the wall. Vessels of any size of any organ system can involve. The disease depends on type of inflammation, the size of blood vessel segment, and the organ. Secondary vasculitis can result from other autoimmune disease, malignancy, allergies etc. ANCA are directed against granular and lysosomal constituents of neutrophils and monocytes. Two fluorescence pattern —a cytoplasmic ANCA (C-ANCA) and a perinuclear ANCA (P-ANCA) staining pattern.
The target antigen of C-ANCA is proteinase-3 (PR3), and of P-ANCA is myeloperoxidase (MPO). Factors contributing to the etiology (blue) and pathogenesis (red). B cells also have a role in the pathogenesis of AAV, primarily through the production of ANCAs. Cyclophosphamide & Rituximab, (chimeric monoclonal antibody against B cells) thus works.T lymphocytes : granulomatous inflammation in WG with IgG1 and IgG4 subclasses, indicates involvement of T-cell, thus MMF works. CD, cluster of differentiation; CTLA4, cytotoxic T-lymphocyte antigen 4 gene; IL, interleukin; PTPN22, protein tyrosine phosphatase, non-receptor type 22 gene.
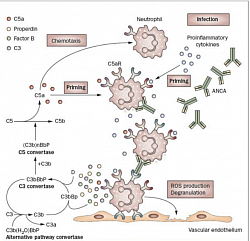
Cellular & complement pathways in AAV: ANCAs stimulate primed neutrophils to undergo a respiratory burst and degranulate. It directly damage the endothelium. It activates the alternative complement pathway with generation of the powerful C5a. C5a and the neutrophil C5aR amplifies ANCA-mediated neutrophil activation, results in necrotizing inflammation of the vessel. Also, according to their cytokine profile and function, CD4+ T helper (TH) cells were originally classified as TH1 or TH2 cells. TH1 cells mainly exert cellular immune responses, and TH2 cells inhibit TH1 responses and participate in humoral immunity. A shift in the TH1-TH2 balance, from a TH1 response to a TH2 response, causes progression of generalized WG.
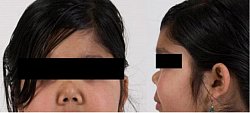
Saddle nose of Wagener Granulomatosis Currently known as Granulomatosis with polyangiitis (GPA). Granulomatous vasculitis that involves the sinopulmonary system but can involve any organ system. Presenting features may include purpuric rash, recurrent sinusitis, epistaxis, shortness of breath, and/or hemoptysis from alveolar hemorrhage and necrotizing glomerulonephritis with significant renal impairment, have a positive ANCA finding in 90% of cases, (c-ANCA, or anti-PR3).
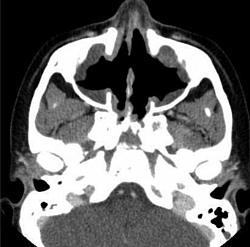
Paranasal sinuses showing erosion & loss of sinus wall.Criteria for GPA include 3 of the following 6 features: 1.hematuria and/or significant proteinuria, 2. Granulomatous inflammation on biopsy (shows necrotizing pauci-immune glomerulonephritis.) 3. Nasal sinus inflammation 4. Subglottic, tracheal, or endobronchial stenosis, 5. Abnormal chest radiography or CT findings, 6. PR3 ANCA or c-ANCA staining in WG.
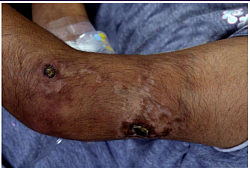
Tender erythematous nodules in Cutaneous Polyarteritis Nodosa (PAN).Polyarteritis nodosa is a necrotizing vasculitis of medium-sized arteries. Features include vasculitic skin lesions, hypertension, Microscopic polyangiitis (MPA) is a necrotizing vasculitis associated with glomerulonephritis and pulmonary capillaritis. Presenting features include purpuric rash, proteinuria and/or hematuria, hemoptysis, CNS involvement, and arthralgias. [7] MPA is associated with ANCA, most commonly with a perinuclear pattern (p-ANCA) and antibodies against myeloperoxidase (anti-MPO).
Necrotizing nodules of Cutaneous Polyarteritis Nodosa. neuropathy, and myalgia. Note that renal involvement does not manifest as glomerulonephritis, as occurs with small-vessel disease. Skin involvement (eg, livedo reticularis, tender subcutaneous nodules, other vasculitic lesions).
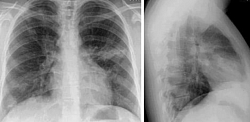
Chest x-ray of pulmonary infiltrates in Churg Strauss Syndrome (CSS). Churg-Strauss syndrome (CSS) is an eosinophilic granulomatous vasculitis characterized predominantly by pulmonary involvement. Patients typically have a previous history of asthma, allergic rhinitis, and/or sinusitis. A characteristic feature is the finding of nonfixed pulmonary infiltrates.
4. Other Vasculitis
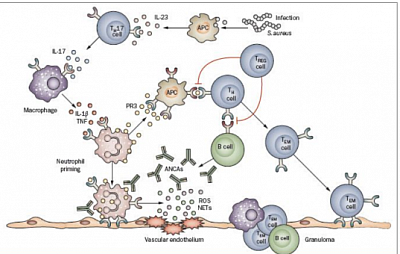
ANCA-mediated neutrophil activation induces the generation of neutrophil extracellular traps (NETs) containing PR3 and MPO. NETs have several pathogenic roles in AAV—they can adhere to the endothelium causing damage, and they might also activate plasmacytoid dendritic cells and B cells to produce interferon and ANCAs, respectively
___________________________________
Takayashu disease
Transmural inflammation and evidence of intramural giant cells. It involves the aorta and its major branches. Characteristic clinical features are caused by stenotic large vessels and subsequently decreased blood supply to the organ systems. Classically, children present with claudication, absent peripheral pulses, blood pressure abnormalities, strokes, and features of internal organ ischemia.
Classification criteria:angiographic abnormalities (conventional, CT, or MRI) of the aorta or its main branches (mandatory criterion), plus at least one of the following features: Decreased peripheral artery pulse and/or claudication of extremities. Blood pressure difference of more than 10 mm Hg. Bruits over aorta and/or its major branches. Hypertension (related to childhood normative data)
Kawasaki
Classification criteria for Kawasaki disease include a fever persisting for at least five days (mandatory criterion) plus 4 of the following 5 features: Changes in peripheral extremities or perineal area, Polymorphous exanthema, Bilateral conjunctival injection, Changes of lips and oral cavity (injection of oral and pharyngeal mucosa), Cervical lymphadenopathy.
Henoch-Schönlein Nephritis (HSN)
Classification criteria for Henoch-Schönlein purpura include palpable purpura (mandatory criterion) in the presence of at least one of following 4 features:
Diffuse abdominal painAny biopsy finding that reveals predominant lgA depositionArthritis or arthralgia (arthritis is acute in any joint)Renal involvement (any hematuria, proteinuria)involves vessels of all sizes. The diagnosis is made clinically in patients with recurrent oral ulcers who also have recurrent genital ulcers, uveitis, various skin lesions, and/or a positive pathergy test.
Anti-GBM antibody disease/Goodpasture syndrome
is a type of vasculitis caused by deposition of anti-GBM antibodies in small vessels of lungs and kidneys. Goodpasture syndrome clinically manifests with rapidly progressive glomerulonephritis and/or pulmonary hemorrhage (pulmonary renal syndrome).
_________________________________
5. Pathophysiology
ANCA antibodies are directed towards cytoplasmic proteins within neutrophils and monocytes (eg, PR3, MPO), which may also be expressed at the cell surface, particularly on stimulated cells. In vitro studies have shown that ANCA IgGs can directly activate neutrophils and monocytes by both Fc receptor engagement and direct Fab2 binding to antigen. These activated cells interact with endothelial cells via adhesion molecules and release inflammatory mediators, such as toxic granule enzymes and reactive oxygen metabolites that cause apoptosis and necrosis. In addition, anti-MPO IgG may activate MPO itself triggering an oxidative burst and resulting in severe endothelial damage. ANCA-activated neutrophils may release factors that activate the alternative complemen.
____________________________________
6. Aetiologic factors
Genetic Factors
HLA genes located on the short arm of chromosome 6, which are also associated with many autoimmune diseases. HLA-DPB1*0401, HLA-DR4 and HLA-DR13(6) are associated with WG. HLA-DRB4 might be a genetic risk factor for CSS, and HLA-DRB1*0901 to be associated with MPA.
Silica
Silica dust exposure is associated with a variety of autoimmune disorders, including AAV, which is thought to evoke an immune response and inflammatory reactions. Accelerated apoptosis of polymorphonuclear leukocytes and macrophages might act as a trigger in the development of AAV. Silica can activate alveolar macrophages, stimulate lymphocytes, attract neutrophils, and induce inflammation and activation of fibroblasts. MPO released by neutrophils is taken up by alveolar macrophages and presented to immunocompetent cells, resulting in the development of anti-MPO autoantibodies.
Bacterial Infections
Bacterial infections have been associated with the initiation and relapse of WG. Among these bacteria, S. aureus seems to have the strongest association with WG. Superantigens from S. aureus might stimulate B cells and T cells, leading to AAV development. In addition, cell-wall components of S. aureus might polyclonally activate B cells, which results in the persistent expression of ANCAs. Another hypothesis is that S. aureus might directly prime neutrophils, which leads to membrane expression of PR3; primed neutrophils can be fully activated by anti-PR3 autoantibodies, resulting in respiratory burst and degranulation with induction of tissue damage.
Drugs
A variety of drugs can cause AAV, including propyl-thiouracil (the most common cause of drug-induced AAV), hydralazine, D-penicillamine and minocycline. It is thought that MPO might undergo structural change following interaction with propylthiouracil and could then stimulate T cells to activate B-cell production of ANCAs. In addition, drugs such as sulfasalazine induce neutrophil apoptosis, which can promote autoantibody production by increasing expression of ANCA target antigens on the surface of apoptotic neutrophils.
__________________________________
7. Management (Agents used in Vasculitis)
IMMUNO-MODULATORY
1. Intravenous immunoglobulin (IVIG)
May absorb superantigens or toxins in Kawasaki disease. May saturate available Fc receptors. May block cytokines, cytokine receptors, or both. May absorb complement activation products. May down-regulate immunoglobulin synthesis. Blocks Fc receptors on macrophages. Suppresses inducer T and B cells and augments suppressor T cells. Blocks complement cascade. May increase CSF IgG (10%).
IMMUNOSUPPRESSIVE
2. Azathioprine
Works primarily on T cells. Suppresses hypersensitivities of cell-mediated type and causes variable alterations in antibody production. Immunosuppressive, delayed hypersensitivity, and cellular cytotoxicity tests are suppressed to a greater degree than antibody responses.
3. Cyclophosphamide (Cytoxan)View full drug information
Cyclic polypeptide that suppresses some humoral activity. As an alkylating agent, the mechanism of action of the active metabolites may involve cross-linking of DNA, which may interfere with growth of normal and neoplastic cells.
4.Mycophenolate mofetil (CellCept); Mycophenolic acid (MyFortic)
Inhibits inosine monophosphate dehydrogenase and suppresses de novo purine synthesis by lymphocytes, thereby inhibiting their proliferation. Inhibits antibody production.
Two formulations are available and are not interchangeable. The original formulation, mycophenolate mofetil (MMF, Cellcept) is a prodrug that once hydrolyzed in vivo, releases the active moiety mycophenolic acid.
5. Prednisolone
Used to control acute symptoms and laboratory evidence of inflammation. May decrease inflammation by reversing increased capillary permeability and suppressing PMN activity. Stabilizes lysosomal membranes and also suppresses lymphocytes and antibody production.
ANTI B LYMPHOCYTES
6. Infliximab (Remicade)
Chimeric IgG1k monoclonal antibody that neutralizes cytokine TNF-alpha and inhibits its binding to TNF-alpha receptor. Reduces infiltration of inflammatory cells and TNF-alpha production in inflamed areas.
7. Rituximab (Rituxan)
Chimeric IgG1-kappa monoclonal antibody directed against the CD20 antigen found on the surface of normal and malignant B lymphocytes. The Fab domain binds to CD20 antigen on B lymphocytes, and the Fc domain recruits immune effector functions to mediate B-cell lysis in vitro. Possible mechanisms of cell lysis include complement-dependent cytotoxicity (CDC) and antibody-dependent cell-mediated cytotoxicity (ADCC).
8. Methotrexate
Antimetabolite that inhibits dihydrofolate reductase, thereby hindering DNA synthesis and cell reproduction. Effects may also be mediated by adenosine via the inhibition of aminoimidazole carboxamide ribonucleotide (AICAR) transformylase, leading to increased release of adenosine. Adjust dose gradually to attain satisfactory response.
Anticoagulant Class
These agents provide immediate and long-term treatment of vascular thrombosis.
Augments activity of antithrombin III and prevents conversion of fibrinogen to fibrin. Does not actively lyse but is able to inhibit further thrombogenesis. Prevents reaccumulation of clot after spontaneous fibrinolysis.
Provide as continuous heparin infusion to maintain aPTT at 1.5 times the control.
Enoxaparin
Low molecular weight heparin. Augments activity of antithrombin III and prevents conversion of fibrinogen to fibrin. Does not actively lyse but is able to inhibit further thrombogenesis. Prevents reaccumulation of clot after spontaneous fibrinolysis.
Advantages include intermittent dosing and decreased requirement for monitoring. Heparin anti–factor Xa levels may be obtained if needed to establish adequate dosing.
Warfarin (Coumadin)
Interferes with hepatic synthesis of vitamin K–dependent coagulation factors.
Antibiotics
Prophylaxis for P jiroveci with cyclophosphamide therapy.
As P jiroveci prophylaxis for patients taking cyclophosphamide. Dihydrofolate reductase inhibitor in combination with sulfonamide.
___________________________________
8. Investigation
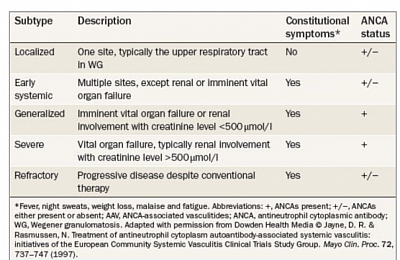
Advance therapy: European Vasculitis Study Group (EUVAS) has proposed to classify AAV according to particular subtypes in order to assign different treatment regimens.
Laboratory Studies
1. General laboratory testsCBC count and differential may reveal normochromic, normocytic anemia; leukocytosis; thrombocytosis consistent with inflammatory process; and eosinophilia in Churg-Strauss syndrome.
C3 and C4 levels are usually elevated.
BUN, creatinine, liver enzyme levels may be abnormal, depending on involvement.
Urine studies for hematuria and/or proteinuria are indicated.
Lumbar puncture may reveal pleocytosis, elevated protein levels.
Antineutrophil cytoplasmic antibody (ANCA) is indirect immunofluorescence to detect staining pattern, either cytoplasmic proteinase 3 (PR3) or perinuclear myeloperoxidase (MPO) by enzyme-linked immunoassay (ELISA) ANCA positivity in 90% with granulomatosis with polyangiitis (GPA) (formerly Wegener granulomatosis), 79% of who are cytoplasmic.
PR3-ANCA and MPO-ANCA positivity have a high sensitivity and specificity for the diagnosis of GPA and microscopic polyangiitis (MPA), respectively.
The association of Churg-Strauss syndrome and ANCA positivity is approximately 40%.
Atypical ANCA finding is nonspecific in infection and inflammatory bowel disease.
Anti-glomerular basement membrane (GBM) antibody testing is indicated for pulmonary renal syndromes.
Bacterial - Mycoplasma PCR and serology, antistreptolysin O test (ASOT), syphilis serology, Mantoux skin test
Viral - Serology for hepatitis B and C, parvovirus B19, HIV, herpes simplex virus, Epstein-Barr virus (EBV), cytomegalovirus (CMV), varicella.
Antiphospholipid antibody syndrome studies include the following:
lupus anticoagulant, Anticardiolipin antibody by ELISA, Anti-β2-microglobulin-1 antibody by ELISA.
Conventional angiography generally provides information about blood flow, clot formation, and collateral blood flow; whereas CT and/or magnetic resonance (MR) angiography provide visualization of vessel wall thickness and fragility, aneurysm formation, and overall disease activity.
End-Organ Imaging, Chest radiography
This is indicated to screen for pulmonary involvement in granulomatosis with polyangiitis (formerly Wegener granulomatosis), Churg-Strauss syndrome, microscopic polyangiitis, Takayasu arteritis (TA), and Behçet syndrome.
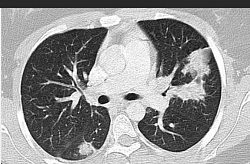
In Churg-Strauss syndrome, nodules, ground-glass opacification, bronchial wall thickening or dilatation, consolidation, septal thickening, and tree-in-bud patterns.
Echocardiography is indicated to assess for coronary artery involvement, especially in Kawasaki disease (KD).
Angiography (CT, MR, conventional) of the aorta and its main branches is indicated when investigating for Takayasu arteritis.
The European Union League Against Rheumatism (EULAR) recommends MRI as a first imaging test for Takayasu arteritis.
Vascular imaging in polyarteritis nodosa (PAN)
Pulmonary function tests – Both obstructive and restrictive patterns seen in pulmonary vasculitis
Electrocardiography – To identify signs of myocarditis or pericarditis
Nerve conduction studies – In patients with peripheral nerve involvement
Electroencephalogram – In patients with CNS involvement
Bronchoscopy/lavage – May be required in patients with lung involvement
Tissue biopsy may be necessary to confirm a diagnosis of vasculitis in systemic and cutaneous polyarteritis nodosa (PAN), ANCA vasculitides, childhood PACNS, and infrequently in Henoch-Schönlein purpura.
Henoch-Schönlein purpura - Skin and/or renal biopsy
Polyarteritis nodosa - Skin biopsy
ANCA-vasculitides - Sinus, renal, lung, and/or skin biopsy
Childhood PACNS - Lesional brain biopsy often needed in small-vessel CNS vasculitis
____________________________________
9. Disease specific treatment
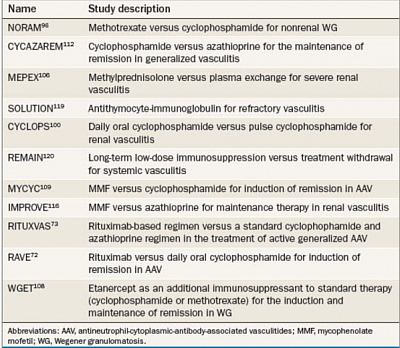
Advances in the Treatment of AAV: The prognosis of untreated AAV is very poor. Major clinical trials in AAV.
The cornerstone of treatment for AAV is corticosteroids in combination with daily oral cyclophosphamide, which has improved the prognosis of patients with AAV dramatically; approximately 90% of patients on this regimen achieve remission. Patients with AAV are treated according to the stage and activity of the disease.
Induction of Remission
Maintenance of Remission
Induction of Remission
Long-term use of oral cyclophosphamide is associated with severe adverse effects, including secondary infection, bone marrow suppression, hemorrhagic cystitis and bladder carcinoma. Trimethoprim-sulfamethoxazole is recommended for prophylaxis of Pneumocystis jiroveci infection during oral cyclophosphamide treatment. Incidence of bladder cancer after first exposure to cyclophosphamide was 5% at 10 years and 16% at 15 years. To obviate this alternative immunosuppressive regimens, such as methotrexate, intravenous cyclophosphamide and rituximab, have been used in major studies.
Methotrexate in Early Systemic AAV.
NORAM study, was initiated by EUVAS
'Early systemic' AAV refers to patients without renal or imminent vital organ failure. No significant difference in remission rates was observed between the two groups; however, the relapse rate at 18 months was significantly higher in the methotrexate group than in the cyclophosphamide group (69.5% versus 46.5%, P=0.023). Regarding adverse effects, leukopenia was less frequent, and liver dysfunction more frequent, in the methotrexate group than in the cyclophosphamide group. On the basis of the results from the NORAM study, EUVAS and EULAR recommend methotrexate as an alternative to cyclophosphamide for induction of remission in non-organ-threatening or non-life-threatening AAV.
Induction of Remission in Generalized AAV.
Two randomized controlled trials of patients with systemic vasculitis (n = 54) and WG (n = 50), respectively, compared the efficacy and safety of daily oral cyclophosphamide versus pulse intravenous cyclophosphamide for the induction of disease remission. Both regimens were associated with a similar rate of remission, but intravenous cyclophosphamide therapy resulted in a lower cumulative dose and less adverse effects.
CYCLOPS:
A randomized controlled trial by EUVAS, compared pulse cyclophosphamide with daily oral cyclophosphamide for the induction of remission of AAV. Relapses were more frequent following pulse therapy than oral therapy, although the study was not powered to detect significant differences in relapse rate; therefore, oral cyclophosphamide can still be considered for use in patients with AAV who experience frequent relapses.
Rituximab
has been tested in open-label clinical trials for the treatment of patients with AAV, with a range of remission rates from 70% to 100%. Randomized controlled trials of rituximab for induction of remission in AAV include the RITUXVAS and RAVE studies.
RITUXVAS trial
compared a regimen of cyclophosphamide plus glucocorticoids (n = 11) with cyclophosphamide plus glucocorticoids and rituximab (n = 33) for induction of remission in generalized AAV (both regimens were followed by maintenance therapy with azathioprine). The rituximab group received four infusions of rituximab 375 mg/m2 plus two infusions of intravenous cyclophosphamide 15 mg/kg. The cyclophosphamide group received between six and ten infusions of cyclophosphamide 15 mg/kg. The study's primary end point—sustained remission at 12 months— was achieved by 76% of patients in the rituximab group and by 82% of patients in the cyclophosphamide group.
RAVE study
compared rituximab to oral cyclophosphamide for induction of remission in AAV. Assigned to receive corticosteroids plus either rituximab (375 mg/m2 per week for four weeks; n = 99) or cyclophosphamide (2 mg/ kg per day; n = 97). Complete remission was acheived in 64% of patients in the rituximab group and 53% in the cyclophosphamide group. Furthermore, rituximab was more effective than cyclophosphamide for induction of remission in the subgroup of patients with relapsing disease (n = 101).However, the role of rituximab in localized disease and in maintenance of disease remission remains to be defined.
Plasma Exchange in Life-threatening AAV.
A trial from EUVAS, termed MEPEX, evaluated the effectiveness of plasma exchange in addition to standard immunosuppression with methylprednisolone in patients with severe AAV with severe acute renal failure. A total of 137 patients with a new diagnosis of AAV (and,, serum creatinine level >500 µmol/l) were randomly assigned to receive seven plasma exchanges (n = 70) or three infusions of 1 g methylprednisolone pulse therapy (n = 67).Both groups also received oral prednisolone and cyclophosphamide. At 3 months of follow-up, renal recovery rate was significantly higher in the plasma exchange group than in the intravenous methylprednisolone group (69% versus 49%, P = 0.02). Furthermore, at 12 months of follow-up, plasma exchange was associated with a 24% reduction in the risk of end-stage renal disease in comparison with intravenous methylprednisolone treatment. Patients with ominous histologic findings can also benefit from plasma exchange for renal recovery. In the group receiving methylprednisolone, the risk of dying from therapy exceeded that of dialysis independence when the proportion of normal glomeruli in the renal biopsy reached a value of <18%. By contrast, a similar risk was noted in the group receiving plasma exchange only when the proportion of normal glomeruli reached a value of <2%.
Other Agents in AAV:
WGET (Wegener's Granulomatosis Etanercept trial), etanercept was used as an additional therapy to the standard regimen (glucocorticoids plus cyclophosphamide or methotrexate) for the induction and maintenance of remission. no significant difference between the two groups at 27 months of follow-up. Solid cancers developed in six patients in the etanercept group, as compared with no such malignancies in the control group.
Etanercept, therefore, is considered ineffective when added to standard therapy for induction and maintenance of remission in WG.
MYCYC trial by EUVAS,
comparing mycophenolate mofetil (MMF) versus cyclophosphamide for induction of remission in AAV, has been initiated.
ADVOCATE study
AVAGOPAP orally-administered, selective complement C5a receptor inhibitor CCX168 (avacopan) in IN 03ducing and sustaining remission in patients with anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV).
Maintenance of Remission of AAV
Although there are remission, relapses in AAV are associated with subsequent progression to end-stage renal disease. Maintenance therapy is crucial to reduce relapses once remission is achieved.
The original regimen for maintenance of remission was oral cyclophosphamide. It causes a variety of adverse effects.
Less-toxic drugs for maintenance of remission, are azathioprine, methotrexate, leflunomide or MMF.
CYCAZAREM study by EUVAS
Either azathioprine (2 mg/kg per day; n = 71) or to continue taking cyclophosphamide (1.5 mg/kg per day; n = 73).
Both treatment groups continued to receive prednisolone (7.5 mg per day),suggesting that azathioprine is as effective as cyclophosphamide for maintaining remission in AAV. Adverse events were comparable between the two treatment groups.
French Vasculitis Study
Patients with WG or MPA were randomly assigned to receive either oral azathioprine (2 mg/kg per day; n = 63) or methotrexate (0.3 mg/kg per week, progressively increased to 25 mg per week; n = 63) for 12 months after induction therapy. No significant difference was found between the two groups in terms of relapse-free survival and toxicity at a follow-up period of 29 ± 13 months.
WG RCT
WG after remission, with cyclophosphamide and prednisolone, were randomly assigned oral leflunomide (30 mg per day) or oral methotrexate (starting with 7.5 mg per week and reaching 20 mg per week after 8 weeks) for 2 years. Leflunomide was more effective in preventing relapses than oral methotrexate but higher rate of adverse events. However, this study was underpowered.
MMF for remission
IMPROVE study
RCT by EUVAS, MMF with azathioprine for maintenance of remission in renal vasculitis. Preliminary results suggest that MMF is less effective than azathioprine; therefore, azathioprine and methotrexate are the preferred drugs for maintenance of remission in AAV.

![Tender erythematous nodules in Cutaneous Polyarteritis Nodosa (PAN).Polyarteritis nodosa is a necrotizing vasculitis of medium-sized arteries. Features include vasculitic skin lesions, hypertension, Microscopic polyangiitis (MPA) is a necrotizing vasculitis associated with glomerulonephritis and pulmonary capillaritis. Presenting features include purpuric rash, proteinuria and/or hematuria, hemoptysis, CNS involvement, and arthralgias. [7] MPA is associated with ANCA, most commonly with a perinuclear pattern (p-ANCA) and antibodies against myeloperoxidase (anti-MPO).](images/th/sd_5f424150e5114.png?no_cache=1598184931)