CKD Renoprotective medicines, Non-immune drugs, MBD, Non-drug management, Diet.
Contents
Section A. CKD MEDICINES
- ANAEMIA MANAGEMEMT
- ACEI & ARB
- MRA
- Renin inhibitor
- Control of BP, Lipid, Glucose
- Bicarbonate
- CaCO3 additional benefit
- Other oxalate scavengers
- Ascorbic acid
- Anti-oxidative stress
- FGF 23 & CKD progression
- Phosphate binders
- PTH suppression
- Renal Bone disease
- TPDI & Post TPDI protocol
Section B. CKD PROTECTION MEDICINES
- Dietary sodium
- Glycaemic control
- New agents for glycaemic control
- Antihypertensives
- Vitamin D
- Endothelin antagonist
Section C. CKD DIET, NON-DRUG MANAGEMENT
- CKD ADVICE,
- Diet
- Lifestyle modifications
____________________________________
Section A: CKD, renoprotective management
Contents
A1. Anaemia management
A2. ACEI & ARB
A3. MRA
A4. Renin inhibitor
A5. Control of BP, Lipid, Glucose
A6. Bicarbonate
A7. CaCO3 additional benefit
A8. Other oxalate scavengers
A9. Ascorbic acid
A10. Anti-oxidative stress
A11. FGF 23 & CKD progression
A12. Phosphate binders
A13. PTH suppression
-
Vitamin D, if level of serum 25(OH) Cholecalciferol is less than 25.
- Alfacalcidol / Calcitriol
- Calcimimetic cinacalcet (Medical parathyroidectomy) in tertiary HPTH
- Paracalcitriol and doxarcalciferol
- Etelcalcetide in secondary HPTH
A14. Renal bone disease
A15. TPDI & Post TPDI protocol
____________________________________
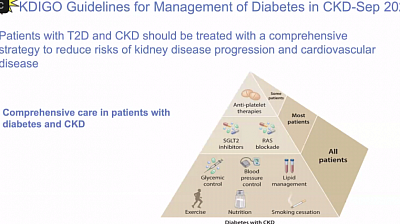
In addition to the specific drugs for primary treatment of DM, hypertension, and other etiological factors, the following medicines are used:
A1. ANAEMIA MANAGEMENT
Anemia (target Hb 10-12g/dl) -
a) Erythropoietin: 60-200 u/kg/week.
b) Iron supplements (po/iv, target TS >25%).
c) New medicine for Anaemia in ESRD: Roxadustat is a first-in-class orally administered inhibitor of hypoxia-inducible factor (HIF) prolylhydroxylase that corrects anemia by a mechanism of action that is different from that of erythropoiesis-stimulating agents (ESAs). As a HIF-PH inhibitor, roxadustat activates a response that occurs naturally when the body responds to reduced oxygen levels in the blood. The response activated by roxadustat involves the regulation of multiple, complementary processes to promote erythropoiesis and increase the blood’s oxygen-carrying capacity. 20,50,100 mg 3 times a week in place of EPO, recently approved in Japan.6. Medicines used in ESRD
____________________________________
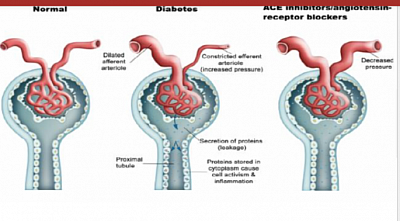
A2. Angiotensin-converting-enzyme inhibitors or Angiotensin-2-receptor antagonists
Use maximum tolerable dose. If high K is a problem, use anti high-K-medicine and continue ACEI or ARB.
____________________________________
A3. Mineralocorticoid receptor antagonist (MRA)
- Steroidal - Spironolactone and Eplerenone.
- Non-steroidal (Finrenone 10 & 20 mg) - new agent passed phase 3 trial. This can be used in low dose with ACEI or ARB. Hyperkalaemia is less likely than the steroidal MRA).
____________________________________
A4. Aliskerin - Renin antagonist. Do not use in CKD-3. Contraindicated because of fatal hyperkalaemia. It can be used in NON-CKD cases of hypertension with eGFR >60.
____________________________________
A5. BP control, lipid control and DM control need to be optimized according to the guidelines.
____________________________________
A6. Sodium bicarbonate tablets - you need to keep HCO3 level above 22. Acidosis primes renal fibrosis.
____________________________________
A7. CaCO3 (additional benefit) - Oxalate from oxalate rich food (10% supply of urinary oxalate) reaches kidney and contributes to crystal nephropathy (oxalate nephropathy) causing progression of interstitial fibrosis with incidences of AoCKD and fast progression to ESRF. Calcium carbonate phosphate binders also bind with food oxalate and reduces it's absorption. Reduces CKD progression.
____________________________________
A8. Other oxalate-scavengers -
Endogenous oxalate (90% suppliers of urinary oxalate) from amino acid metabolism can be reduced by low dose pyridoxine. You can try for 3 months. Can continue if response is good. Dietary oxalate can be reduced by calcium supplements as mentioned above and with probiotic. Probiotic degrades oxalate in the gut. You can try for 3 months. Can continue if response is good.
____________________________________
A9. Ascorbic acid - is metabolized to oxalate. This way Vitamin C and citrus fruits can fasten CKD progression. Avoid these in your patients.
____________________________________
A10. Oxidative stress and chronic inflammation - CKD is associated with chronic inflammation, endothelial dysfunction, platelet activation and oxidative stress. CKD progression can be reduced by addressing these.
Statin - Atorvastatin has been shown to reduce endothelial dysfunction in CKD. Use it in hyperlipidemia.Allopurinol - by it's non-xanthine oxidase inhibition action. It inhibits lipid peroxidase (produces oxygen radicals), thus inhibit oxidative stress. Reduces CKD progression. If you get hyperuricemia by lab results, use it. Not much evidence to support this notion. Need further RCTs.Notes on allopurinol & HLA-B*58-01
In Chinese, Thai & Korean, Allopurinol associated drug reaction has been closely associated with HLA-B*58-01, (80-85% in positive cases while only 15_20% in negative cases). HLA-B*58-01 is common in these population than rest of Asian and European population. HLA-B*58-01 genotyping (a codominant allele) could considerably reduce the occurrence of allopurinol-induced SCARs (severe cutaneous allergic reactions) and related deaths. Cost-effectiveness analysis of treating patients with chronic gout (without additional risk factors like CKD3) in Singapore and in Portugal found that HLA-B*58:01-guided ULT (urate lowering therapy) was not cost-effective at this time.
FDA recommended gene testing in susceptible population (Chinese, Korean, Thai) only before starting allopurinol.
c. Febuxostat
A large study of cardiovascular events in gout patients taking one of two medications to prevent excess build-up of uric acid has found that one of the drugs, febuxostat (Uloric, Takeda), increased the risk of death for those with heart disease compared with the other drug, allopurinol. Yet the two drugs were comparable with respect to a combination of nonfatal heart attack, nonfatal stroke, urgent surgery to treat angina, and death due to cardiovascular causes.
FDA advisory to use with informed consent in patients who are allergic to allopurinol.
____________________________________
A11. FGF23 (optimization of CKD renal bone metabolism) - hyperphosphatemia from CKD is associated with upregulation of Fibroblast Growth Factor (FGF23) from renal paranchyma, also known as phosphaturic hormone. FGF23 is important in embryogenesis responsible for the fibrous tissue in the developing fetus. In CKD upregulation of it could contribute to progressive fibrosis. Control of hyperphosphatemia therefore is important in retardation of progression of CKD. Optimization of phosphate binders and vitamin D are required.
____________________________________
A12. Phosphate binders:
a) Calcium based carbonate (Ca 40%) or acetate ( Ca 25%) with food.
b) Noncalcium based:
i. Lanthanum carbonate (divalent metal like calcium, binds phosphate in food. Longterm study showed deposits in bone, but impact yet uncertain).
ii. Sevelamer hydrochloride (a resin that chelate phosphate. Also chelate lipids, drugs vitamins and other trace elements, as such needs caution for other concomitant medication use).
iii. Magnesium hydroxide and Magnesium Carbonate, singly or in combinations of Calcium carbonate or acetate.
iv. Iron Magnesium Hydroxycarbonate.
All showed good phosphate lowering affects.
____________________________________
A13. PTH Suppression : iPTH more than 25 pmol/l, needs suppression by:
a) Vitamin D, if level of serum 25(OH) Cholecalciferol is less than 25.
b) Alfacalcidol / Calcitriol 0.25 - 1 mcg 3 x/wk.
c) Cinacalcet (calcium mimetics) 25-50 mg daily if S Calcium is high. They mimic cakcium and bind to calcium receptor of PTH secreting clear cell of parathyroid gland. Thus inhibit the cells by negative feedback mechanism.
d) Paracalcitriol and doxarcalciferol: these agents binds to vitamin D receptor of PTH secreting cler cells of parathyroid gland, but do not have the action on calcium absorption.
e) Etelcalcetide: In the end of the spectrum you get tertiary hyperparathyroidism where you need either surgical total parathyroidectomy and deltoid implant (TPDI), or in cases of surgical unavailability, medical parathyroidectomy by Cinacalcet, Paracalcitriol, Doxarcalciferol.
(Quotes from this link about Etelcalcetide): In dialysis patients who cannot take cinacalcet because of vomiting, its derivative peptide Etelcalcetide IV weekly 3 times is used. In 2016, Etelcalcetide was introduced for the treatment of secondary HPT. The peptide etelcalcetide directly agonizes the CaSR, thereby reducing circulating levels of PTH and subsequently calcium (ref). By means of a triweekly bolus injection of the drug, a sufficient plasma level of the drug is attained, enabling the administration during dialysis. This makes drug adherence easier for the ESRD patient. Etelcalcetide has been compared to both placebo as to its precursor Cinacalcet (ref). 50% of the patients who received etelcalcetide showed a decrease of PTH levels to ⩽32 pmol/L. Etelcalcetide was expected to give less side effects as vomiting and diarrhea since it is administrated intravenously and thus bypasses the gastrointestinal tract. Unfortunately, no significant difference in side effects was found when comparing etelcalcetide with cinacalcet, probably due to PTH-receptors in the intestinal tract. A 30% decrease of FGF-23 levels was seen in 80% of the patients using etelcalcetide after 27 weeks. Studies investigating the long-term effects of etelcalcetide on mineral bone disease, overall survival, and cardiovascular events are not available yet. The costs of etelcalcetide are significantly higher than its precursor cinacalcet. Therefore, etelcalcetide should be used as a second in line treatment option, in patients unresponsive to oral calcimimetics or for those patients who fail to adhere properly. Etelcalcetide, like cinacalcet, is only registered for the treatment of dialysis patients, not for those who received a kidney transplant.
____________________________________
A14. Renal bone disease (go to B35 in the link)
___________________________
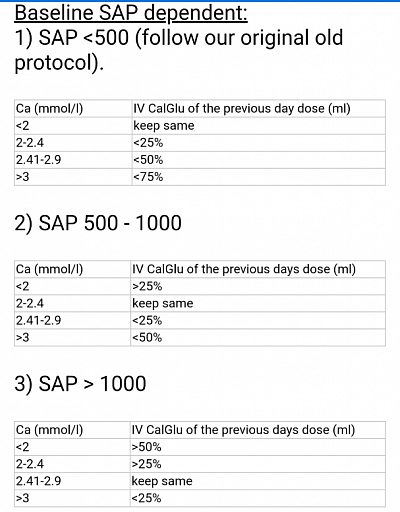
A15. TPDI & Post TPDI protocol: Calcium and active vitamine D replacement
- After Total Parathyroidectomy and Deltoid Implant surgery, iPTH level plummets, so also serum calcium level (corrected for Albumin). Because of Osteoclast hyperplasia, as happened due to hyperparathyroidism with resorption from bones, post parathyroidectomy the calcium starts to be taken up in bones by osteocytes, as a result calcium level also plummets.
- Calcium needs to be replaced by IV Calcium Gluconate (ml= mmol of ca) or IV Calcium Chloride (3x concentrated than gluconate).
- The first dose post TPDI Surgery is half of serum Alkaline Phosphatase (indicative of Osteoclast activity) level. This is knoen as rule of thumb. The IV Calcium needs to be givren diluted to equal volume of NS or D5% over 6 to 12 hours through CVP line.
- Subsequent daily dose depends on daily serum calcium level as per protocol, until the amount comes down to 30 ml. Then oral CaCO3 tablet, maximised dose, along with active Vitamin D in maximum dose to continue. Later both medicines need to be titrated down as required.
- As iPTH level plummets, you need to replace it with large dose initially, and titrate it down subsequently depending on its lrvel. Recommended to begin with Alfacalcufol or Calcitriol 2 to 3 mcg TDS, and titrate it down based on iPTH level.
________________________________
Section B: CKD Non-immune drugs
Contents
B1. Dietary sodium
B2. Diabetic medicines
B3. New agents for diabetes mellitus
B4. Antihypertensives
B5. Vitamin D
B6.Endothelin antagonist
_____________________________________
B1. Dietary Na - reduce salt intake to 5 gm.
- Equivalent to Action of ACEI or ARB in protecting DKD.
- Low-sodium diet (24-h urinary sodium/creatinine ratio (mmol/g) < 121) enhanced the renoprotective and cardioprotective effect of angiotensin receptor blockers (losartan or irbesartan) in type 2 diabetic patients.
_____________________________________
B2. Glycaemic control -
- Results from pancreatic transplant recipients in whom true euglycemia is restored suggest that strict glycemic and metabolic control may slow the progression rate of progressive renal injury even after overt dipstick-positive proteinuria has developed.
- This was also shown in DCCT & UKPDS studies. This is cost effective as well.
_____________________________________
B3. New agents for DM in CKD:
a. Dipeptidyl peptidase inhibitors
- The dipeptidyl peptidase (DPP)-4 inhibitors (ie, gliptins) in type 2 diabetes. Linagliptin, Sitagliptin and Vidagliptin.
- Inhibit the breakdown of the incretin hormones such as glucagon like peptide 1 (GLP-1) secreted by the GI tract in response to food intake, and leads to insulin secretion in a glucose-dependent manner, while decreasing glucagon release.
GLP-1 also slows gastric emptying.
b. Alpha-glucosidase inhibitors
- Alpha-glucosidase inhibitors (acarbose) decrease the breakdown of oligosaccharides and disaccharides into monosaccharides in the small intestine, slowing the absorption of glucose.
- Adverse effects are bloating, flatulence, and abdominal cramping. Minimally absorbed, with less than 2%.
c. Sodium-glucose cotransporter2 (SGLT2) inhibitors.
- Inhibits renal glucose absorption in the proximal tubule, the site in the kidney where approximately 90% of glucose reabsorption occurs.
- This leads to increased excretion of glucose in the urine. Also accompanied by increased urinary excretion of sodium, which helps further blood pressure lowering.
- This explains additional cardiovascular and renal benefits.
- If patient is on insulin, to continue, otherwise there is increased incidence of amputation.
- Even though, it is indicated in patients with eGFR of more than 30ml/min, many nephro-cardiologist across the world use it in moderate CKD with eGFR as low as 16ml/min.
d. Glucagonlike peptide-1 (GLP-1) receptor agonists or incretin mimetics
- The GLP-1 agonists exenatide and liraglutide are also known as injectable incretin mimetics.
- These drugs promote insulin release, delay glucagon release, and slow gastric emptying and are less likely to cause hypoglycemia.
- They may cause pancreatitis and acute renal failure.
e. Amylin analogs
- Amylin is a peptide co-secreted with insulin and is deficient in diabetes. Its actions are complementary to insulin in regulating plasma glucose. Amylin reduces postprandial glucagon.
- Pramlintide - Amilin analog is given as an injection with insulin. Dose adjustment for pramlintide are not required in the presence of mild-to-moderate renal failure.
_____________________________________
Careful blood pressure control is needed to prevent the progression of diabetic nephropathy and other complications; however the optimal lower limit for systolic blood pressure is unclear. In the UKPDS, a 12% risk reduction in diabetic complications was found with each 10 mm Hg drop in systolic pressure, the lowest risk being associated with a systolic pressure below 120 mm Hg. SPRINT study showed similar result in all, but at the expense of increased incidence of hypotension and mortality.
a. ACEI
ACE inhibition has been shown to delay the development of diabetic nephropathy. In the ACE inhibition arm of a large trial, only 7% of patients with microalbuminuria experienced progression to overt nephropathy; however, in the placebo-treated group, 21% of patients experienced progression to overt nephropathy. The beneficial effect of ACE inhibition on preventing progression from microalbuminuria to overt diabetic nephropathy is long-lasting (8 years).
b. ARB
Both DM1 & DM2 are characterized by the appearance of microalbuminuria, which leads to overt proteinuria and progressive loss of GFR. A series of renal biopsy samples from patients with type 2 DM and proteinuria revealed that a significant proportion of these patients had glomerular lesions other than the classic lesions associated with type 1 diabetic nephropathy.
Two studies (the Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan [RENAAL] Study and the Irbesartan Diabetic Nephropathy Trial [IDNT]) demonstrated that angiotensin II receptor blockers (ARBs) are superior to conventional therapy and amlodipine in slowing the progression of overt nephropathy.
All of you agree that the combination is not good. ON-TARGET and NEPHRON-D studies showed that.
d. ALISKIREN.
Contraindicated in eGFR <60 ml/min.Renin antagonist. Do not use in CKD-3. Contraindicated because of fatal hyperkalaemia. It can be used in NON-CKD cases of hypertension with eGFR >60.
_____________________________________
B5. Vitamin D
May have a role in renin inhibition, and vitamin D supplementation may be useful in reducing proteinuria in patients with diabetic nephropathy. Patients with diabetic nephropathy with stage 3 chronic kidney disease (eGFR 59 – 30 mL/min/1.73 m2) or more advanced stages should be evaluated for their vitamin D and parathyroid hormone status as recommended (KDIGO).
_____________________________________
B6. Endothelin Antagonist
Avosentan: Endothelin antagonists have demonstrated antifibrotic, anti-inflammatory, and antiproteinuric effects in experimental studies. It reduces proteinuria and BP when added to ACEI or ARB.
_____________________________________
Section C. CKD DIET & NON-DRUG MANAGEMENT
Section C. CKD DIET & NON-DRUG MANAGEMENT
C1. CKD ADVICE
C2. Diet
C3. Lifestyle modifications
_____________________________________
The pills cannot heal unless you go extra miles with patient education for life long diseases. Go to the following link, and let your patient toggle it anywhere anytime lifelong.
C1. CKD ADVICE (Renal failure advice)
C2. CKD DIET (Food)- the other important thing that goes through mouth othe then drugs, is food. Must be careful. Common items may have problem with kidney issues.
C2.1 Nutrition in Numbers:
- Salt (5gm/day) and required amount of water.
- Furosemide for excess water and Fluid as permitted.
- Weight control with stable BMI<25).
- Dietary control of Blood Pressure.
- Dietary control for diabetic glucose level.
- Controled fruits / vegetables rich in Potassium (<2g/day), Phosphate (<0.8g/day), Oxalate (<0.2g/day) and Ascorbate (converted to Oxalate in Liver.
- Protein intake in CKD-ND 0.8g/kg/day + TUP, in CKD-HD 1.2g/kg/day, in CKD-PD 1.5g/kg/day and in CKD-Tx 0.8g/kg/day. (Normal intake is 1g/kg/day. CKD needs protein restrictions. HD is hypercatebolic, hence requires slightly more. PD is associated with Peritoneal protein loss, hence needs additional amount. Transplant Recipient is itself a CKD, hence follows CKD diet.
- To maintain hemoglobin (Hb) 11-12g/dl, needs iron supplements.
- Needs vitamin supplements and Calcium with food. May also need active Vitamin D.
C3. CKD LIFESTYLE Lifestyle care - kidney is vulnerable to many other factors pertaining to lifestyle. Patient needs to be aware of it.
_____________________________________